Surfacing Patient Anomalies
Using machine learning to identify anomalies in clinical trial data helps clinical data managers prioritize their attention to the most likely issues and identifies patterns pointing to potential problems with trials that can be addressed quickly.
Clinical data managers are responsible for reviewing data being returned from clinical trial sites for accuracy. They are looking for anomalies that might skew the results of a trial that need to be addressed. A machine-learning model with a human-in-the-loop feedback mechanism facilitates faster review of potential anomalies.
A machine learning autoencoder model identified whether a patient had a high-likelihood of being an anomaly.
A data app was built on top of the output of that model to facilitate review by clinical data managers.
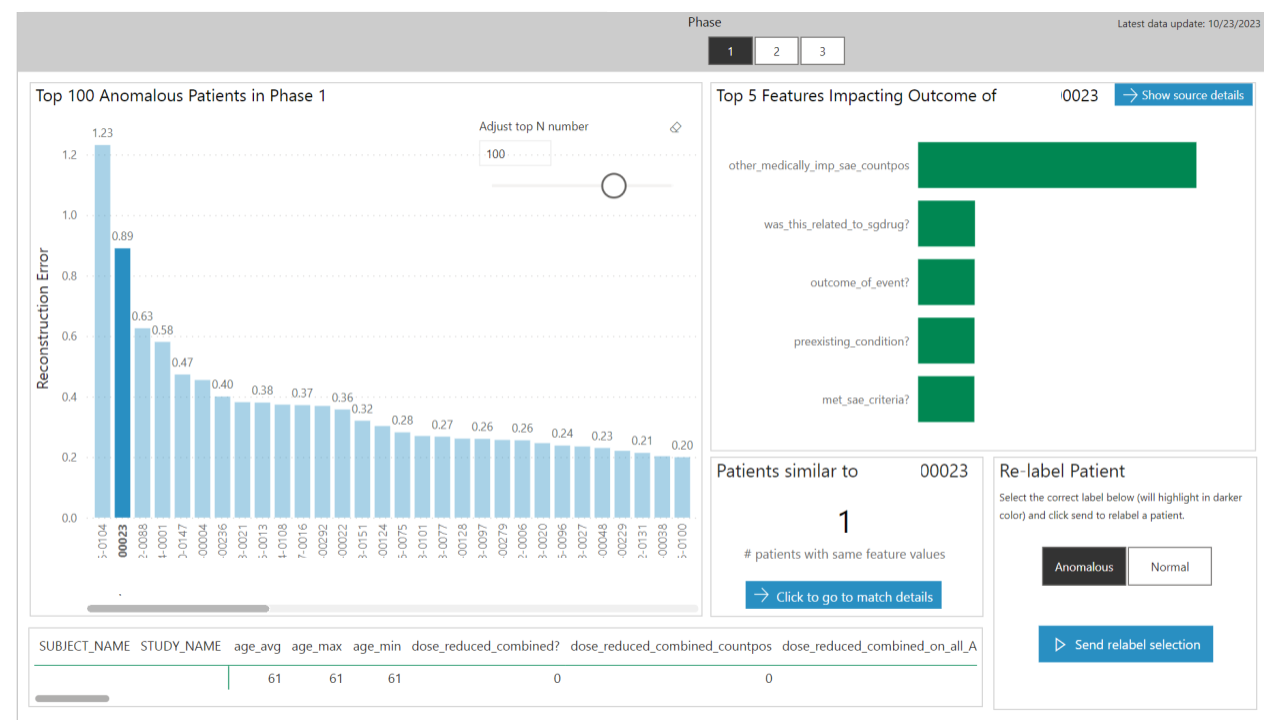
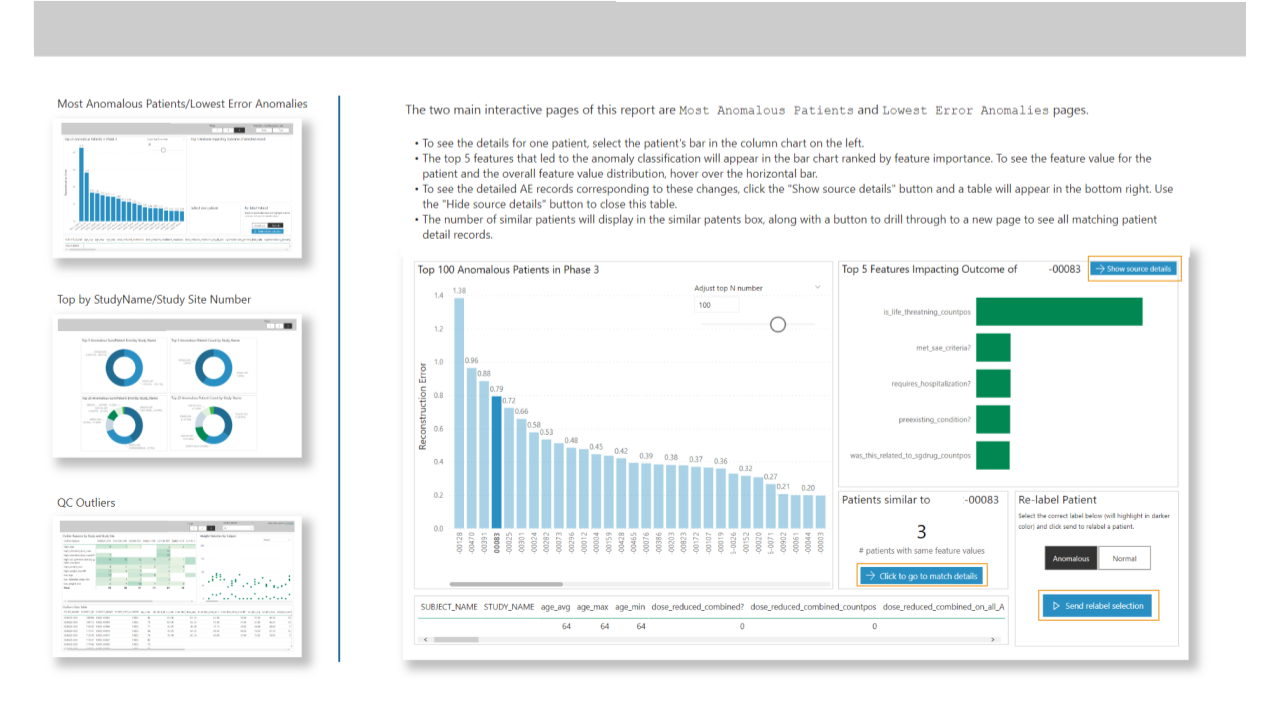
The autoencoder provided a ranking of why it classified a patient as an anomaly, presented to the clinical data manager as a top-N chart, with the ability to see all source details during evaluation.
Incorporating data necessary for clinical review into the app turns this into a single-pane-of-glass app for review of anomalies, reducing the need to switch systems for reporting and action.
After evaluating the output from the model, the clinical data manager could override the predicted outcome and relabel the patient. This was then incorporated into the autoencoder model to help it better classify anomalies.
Distinguishing between outliers and anomalies
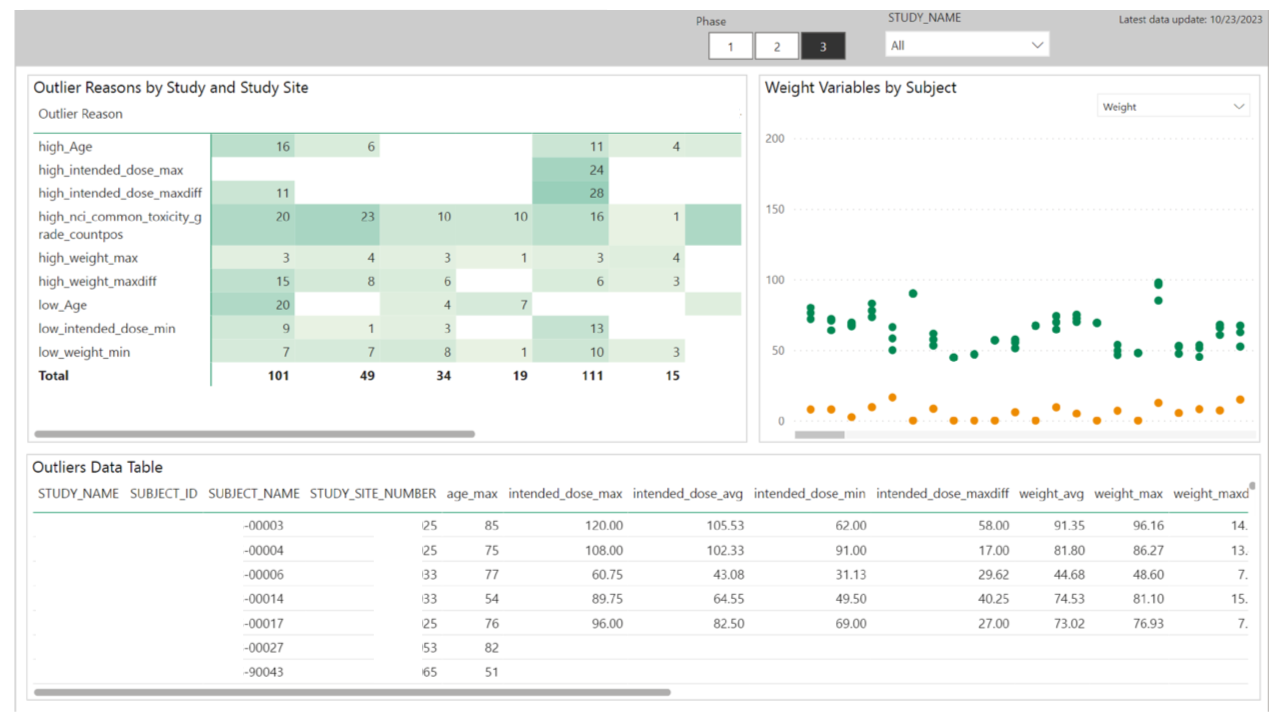
In this context, an outlier is a value that looks like a data quality issue, rather than a medical anomaly. An example may be that a dose is entered as 10.2 when the intended dose range is between 91-108. This is likely a data entry error that should be corrected in the source system for clean data.
Trends in outliers help clinical data managers identify potential issues with specific trials or trial sites. Identifying these early and correcting them saves money and ensures fidelity of clinical trials, a critical element in bringing a life-saving medication through the approval process and to the market.
Supporting behavior change
Clinical data managers historically performed their review in source systems and with varying individual approaches to review. Bringing the data necessary for review into a data application was a change in their workflow.
In-app documentation provides explanation of how to use the application and the distinctions about types of analysis that are available to support their review. Keeping documentation support in the application reduces friction in adoption of the new tool.